In today’s VETgirl online veterinary continuing education blog, we review feline urethral obstruction (FUO) in a 2-part blog. FUO is a common disorder presenting to both the general practitioner and the emergency clinician. In Part 2 today, we review overall treatment, including fluid therapy, emergency drug therapy, sedation and unblocking of the FUO patient.
Treatment
Treatment of the FUO patient is aimed towards aggressive fluid therapy, alleviation of obstruction, emergency drug therapy (e.g., for treatment of electrolyte abnormalities), symptomatic supportive care, analgesia, anti-spasmotics (if necessary), client education, and monitoring.
Fluid therapy
In the FUO patient, aggressive intravenous (IV) fluid therapy is indicated as many patients often have a profound post-obstructive diuresis (i.e., > 2 ml/kg/hour) and may present dehydrated and azotemic. In one study, almost 50% of cats were found to have a post-obstructive diuresis within the first 6 hours of hospitalization (Francis, 2010); this was more likely to develop in patients with a pH < 7.35 (e.g., metabolic acidosis). An initial fluid bolus with a balanced, buffered isotonic crystalloid (e.g., Norm-R, LRS, Plasmalyte, etc.)* at 20-30 ml/kg can be administered over the first hour to help increase perfusion and aid in correction of azotemia and electrolyte abnormalities. This can be initiated during the sedation and unblocking process, and does not need to “wait” until the unblocking has been performed. Following alleviation of the urinary obstruction, fluids should be maintained at 2.5-5X maintenance. The author typically uses 50-60 mls/cat/hour for the FUO patient until the patient is hydrated and the urine is clear.
*NOTE: The use of 0.9% saline has not been shown change the outcome or resolution of hyperkalemia in these critically ill patients (Drobatz 2008); as this fluid does not contain a buffer, the author prefers a buffered isotonic solution instead.
The patient should be assessed carefully to ensure that volume overload does not occur, particularly in patients with cardiopulmonary disease (e.g., gallop, murmur, etc.). Fluid therapy should be continued until azotemia and clinical signs resolve (typically 2-4 days); IV fluids should then be slowly tapered to ensure that the azotemia and polyuria has resolved and the patient can maintain hydration. Due to the severe post-obstructive diuresis that occurs in these cats, fluid therapy should be carefully matched by monitoring ins and outs. Urine output (UOP) should be monitored carefully, particularly in azotemic patients with FUO.
Emergency drug therapy (e.g., hyperkalemia)
For the critically ill FUO patient (e.g., bradycardiac, obtunded, hypotensive), priority should be aimed at treating the life-threatening hyperkalemia. The author prefers the use of calcium gluconate 10% (50-100 mg/kg, IV slow over 10-15 minutes) to help increase the threshold potential (as hyperkalemia lowers the threshold potential) for severe hyperkalemia. Note that calcium does not directly treat the hyperkalemia. Hyperkalemia decreases the resting potential, making it less negative (initially making the cells more hyperexcitable) (Dibartola, see Figure 1 below). The administration of calcium works by increasing the normal threshold membrane potential, thereby normalizing the difference between the two potentials and providing a cardioprotective effect.
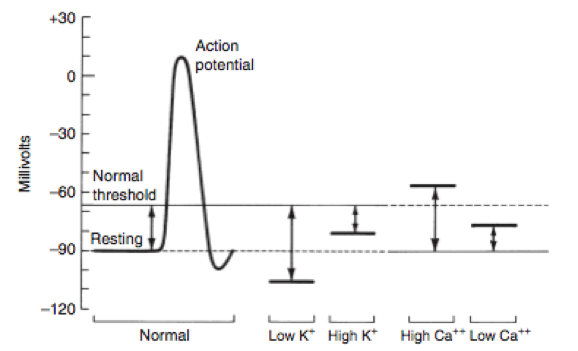
Figure 1. Dibartola’s Fluid Therapy chart on the effect of electrolytes on the action potential. Image from Dibartola SP, Disorders of potassium. In Fluid Therapy in Small Animal Practice, 2000.
Alternatively, the use of sodium bicarbonate (1-2 mEq/L, IV slow over 15-30 minutes) can be considered for severe hyperkalemia and metabolic acidosis. While the combined administration of insulin:dextrose can be considered, the author does not routinely use this method, as profound persistent hypoglycemia (and secondary dextrose administration) can be seen for hours afterwards (which indirectly increases cost of hospitalization due to frequent blood glucose monitoring). This is based on individual clinician preference. Once these treatments have been initiated, the clinician should focus on relieving the urinary obstruction.
Sedation
Sedation protocols should be based on the stability of the patient, the severity of electrolyte abnormalities, and the severity of clinical signs (e.g., bradycardiac, comatosed, obtunded, etc.). Ideally, sedation protocols should be quick acting, reversible, and cardiovascularly-sparing. The author’s preference for sedation for unblocking in a typical, stable FUO case is (Dosages are listed as total cat doses):
• Butorphanol: 4 mg IV total per cat
• Diazepam: 2.5 mg IV total per cat
• Ketamine: 10 mg IV total per cat
In the critically ill, obtunded FUO case, very little sedation – if any at all – may be necessary. In critically ill cases requiring minimal sedation, the author prefers (Dosages are listed as total cat doses):
• Butorphanol: 2 mg IV total per cat
• Diazepam: 1.25 mg IV total per cat
Alleviation of urinary obstruction
Alleviation of the urinary obstruction is dependent on clinician preference. The author prefers using a Tomcat sterile polypropylene catheter initially, followed by a 3.5 to 5 French red rubber catheter. While a variety of catheter types are available (e.g., rigid olive tip, Slippery Sam, MILA, etc.), the author recommends using ones are soft, pliable, and atraumatic. A closed collection system should be used to prevent ascending infection. Aseptic technique should be used as best as possible. In order to alleviate the obstruction, the lubricated urinary catheter should be well seated into the tip of the penis, making sure to pull the prepuce caudally to straighten the penile flexure and aid in passing the urinary catheter. A sterile syringe with saline should be used to copiously flush the urethra, with the goal to dislodge and flush the obstructing materials (e.g., crystals, blood clots, cellular debris, calculi, etc.) out of the urethra (either back into the urinary bladder or antegrade out of the tip of the urethra). The author prefers to flush aggressively as the temporary catheter is removed, followed by immediate placement of a longer indwelling urinary catheter (e.g., red rubber, Slippery Sam). The catheter should be sutured in immediately (e.g., Chinese finger trap, etc.) and the bladder copiously flushed.
In this video here, you can view us placing a MILA urinary catheter in a cat.
Analgesia
The use of coccygeal blocks can be considered for the FUO patient. This will provide analgesia to the perineal region for approximately 1 hour. Epidurals and coccygeal blocks allow us to provide analgesia without affecting motor function. For a step-by-step procedure of how to perform a coccygeal block, the readers are referred to:
Additional analgesic therapy (e.g., buprenorphine 11-22 mcg/kg, IV q 6 OR long-acting buprenorphine in the form of Simbadol™) should be continued while the urinary catheter is in place. The author typically doses Simbadol on lean body weight or at half the labeled dose (e.g., 0.12 mg/kg SQ once a day for 3 days). The use of acepromazine (0.005-0.01 mg/kg, IV) can also be used as an anxiolytic and to aid with urethral spasms (See “anti-spasmotics” below for more information).
Symptomatic supportive care
Additional treatment of the FUO includes:
• Nursing care
• Monitoring of the UCS (e.g., UOP, ins and outs)
• E-collar placement
• Blood pressure monitoring
• Evaluation of hydration status
• Nutritional support (e.g., water should be available at all times, and the author prefers to feed a grueled canned food while hospitalized to help increase water intake)
• Symptomatic supportive care
• Daily renal panel (e.g., electrolytes, BUN, creatinine, PCV/TS)
Once the patient is no longer azotemic, the patient is hydrated, and the urine appears to be clearing up (e.g., free of gross clots, debris, crystals, RBC, etc.), the urinary catheter can be removed (typically 24-72 hours after placement). The author typically flushes the urinary catheter while removing it to help clear the urethra. The patient should be monitored for several hours after UCS removal to ensure adequate urination prior to discharge.
Antibiotics
The use of prophylactic antibiotics for the catheterized FUO patient is debated; the author prefers not to antibiotics while the patient has an indwelling UCS due to the risk of causing a more resistant urinary tract infection or contributing to pyelonephritis. That said, iatrogenic infection can develop as a direct consequence of catheterization, despite our best attempts to use aseptic technique. Ideally, the use of antibiotics should be limited for use in the patient once the catheter has been removed (or pending urine or catheter tip culture). That said, if signs of infection (e.g., fever, pyruria, bacteriuria, etc.), renal pain, or sepsis are present, a urine sample should be obtained for culture and antibiotics initiated promptly (Lane 2012).
Anti-spasmotics
The use of smooth muscle relaxants [such as prazosin (0.25-1 mg/cat PO q 8-12 X 3-10 days) or phenoxybenzamine (2.5-7.5 mg/cat PO q 12-24 hours X 3-10 days)] or striated muscle relaxants (e.g., diazepam, 0.1-0.2 mg/kg IV q. 8-12) can be used in cats with urethral spasming; ideally, oral diazepam should not be used in cats due to the rare risks of acute hepatic necrosis with dosing (intravenous administration is very safe, however). NOTE: The use of very tight-fitting urinary catheters may result in urethral spasming, in the author’s opinion.
Decompressive cystocentesis (DC)
Copper et al have previously published the use of decompressive cystocentesis, along with a “stress-free” environment (e.g., dark room, sedation) as a means of treating the FUO patient. In the author’s opinion, routine sedation, catheterization and alleviation of the obstruction is still key. While Hall et al have shown that the use of DC is safe in the FUO, it should be followed by placement of an indwelling urinary catheter, hospitalization and supportive care. Ideally, the use of DC should not be used as a solitary treatment for cats with FUO due to risk of uroperitoneum and hemoperitoneum.
Long-term management
Owners should be educated on the risk of reoccurrence, the need for potential surgery or nutritional management, litter box husbandry (e.g., n+1 litter boxes, daily scooping, clumping litter, etc.), potential for surgical correction (e.g., cystotomy, perineal urethrostomy, etc.), dietary changes (e.g., to grueled canned food) and lifestyle changes (e.g., minimal stress, etc.). Patients should be thoroughly evaluated for presence of uroliths or crystals, and managed appropriately.
Tips on unblocking:
• Make sure to appropriately sedate prior to catheterization, as adequate sedation will make your unblocking go more smoothly (and less traumatically!)
• Rarely, decompressive cystocentesis may be necessary if the patient is difficult to unblock. This may help relieve backpressure in the urinary tract.
• Appropriate client communication is imperative with FUO. Risks of sedation, risk of bladder rupture, or risk of urethral tear or injury should be discussed with the owner prior to unblocking.
Copyright VETgirl, 2016.
References:
- Lee JA, Drobatz KJ. Characterization of the clinical characteristics, electrolytes, acid-base and renal parameters in male cats with urethral obstruction. J Vet Emerg Crit Care 2003;13(4): 227–33.
- Reineke EL. Feline urethral obstruction: Emergency treatment and stabilization. Western Veterinary Conference 2013, Proceedings.
- Lane IF. Common challenges in feline urethral obstruction. Western Veterinary Conference 2012, Proceedings.
- Francis BJ, Wells RJ, Rao S, et al. Retrospective study to characterize postobstructive diuresis in cats with urethral obstruction. J Feline Med Surg 2010;12:606-608.
- Drobatz KJ, Cole S. The influence of crystalloid type on acid-base and electrolyte status of cats with urethral obstruction. J Vet Emerg Crit Care 2008;18(4): 355–61.
- Cooper ES, Owens TJ, Chew DJ et al. A protocol for managing urethral obstruction in male cats without urethral catheterization. JAVMA 2010;237(11):1261-1266.
- Hall J, Hall K, Powell L, Lulich J. Outcome of male cats managed for urethral obstruction with decompressive cystocentesis and urinary catheterization: 47 cats. JVECC 2015;25(2):256-262.
NOTE: When in doubt, all drug dosages should be confirmed and cross-referenced with a reference guide such as Plumb’s Veterinary Drug Handbook.









Only VETgirl members can leave comments. Sign In or Join VETgirl now!