November 2023
From “oh no” to high flow: Oxygen therapy in veterinary practice
By Melanie Newton, CVT VTS (ECC), Veterinary Technician Supervisor – Emergency Department, BluePearl Veterinary Partners Waltham, MA
In today’s VETgirl blog, guest veterinary technician blogger Melanie Newton, CVT VTS (ECC), Veterinary Technician Supervisor reviews all things oxygen therapy related! Oxygen therapy in veterinary medicine is life-saving in the tachypneic or dyspneic patient. The veterinarian, veterinary technician, and veterinary assistant must know how to delivery oxygen safely to the patient. This may include immediate recognition and administration through flow-by, face mask, oxygen cage, oxygen hood, and different forms of nasal administration.
We’ve all been there. The dog that presents for vomiting and coughing and is diagnosed with aspiration pneumonia. The cat that presents for just “not doing right” and is actually in respiratory distress. We instinctively know these patients need oxygen therapy, but what are the best options for the method of delivery? Our answer depends on the diagnosis, patient, and equipment availability.
DETERMINING THE NEED FOR OXYGEN THERAPY
In some cases, it can be difficult to know when oxygen therapy is warranted. Using both subjective and objective data can help us determine if oxygen therapy is needed and which method of delivery is appropriate for the patient.
Assessing the patient from afar can be very revealing. Prior to even touching the patient, it may be helpful to observe how they are breathing. Some patients can become stressed when manipulated, leading to a falsely elevated respiratory rate. Watching the patient breathe while sitting with their owner or while in the carrier can give you a better idea of how they are truly compensating. Is the patient breathing fast or slow? Does their abdomen seem to be moving with every breath? Is their neck in a neutral position or is it outstretched in an effort to elongate their trachea? Are they resting comfortably next to mom or are they shifting their position frequently? These answers can be obtained in less than a minute, but can give us a great baseline to start.
After carefully observing the patient, performing a quick yet thorough physical exam can help to further determine the patient’s needs. If a patient is struggling to breathe, they may become panicked when restrained. Therefore, a more hands-off approach may be beneficial. Auscultating the heart and lungs should be prioritized. If a temperature is deemed necessary to help rule out causes of the respiratory distress, an aural thermometer may come in handy to help decrease the stress caused by a rectal temperature. Mucous membrane color should be noted, but is not necessarily a reliable indicator of oxygenation status. Pink does not always equal good oxygenation! During the physical exam, flow-by oxygen can be provided as well (and no need to worry if the patient ends up not needing oxygen support. Flow-by oxygen given to a healthy patient over a short period of time will not cause any harm).
When paired with our physical examination findings, a pulse oximeter may give us more information in determining our patient’s need for oxygen support. While obtaining an SPO2 reading is non-invasive, some patients become distressed during this process. If the patient is willing to cooperate for an SPO2 reading, the number may help to determine if oxygen support is needed and then later to tell us if oxygen therapy is helping. Oxygen therapy is indicated in patients with an SPO2 of 93% or lower. However, the saying “treat the patient, not the number” rings true when using a pulse oximeter. Even if the SPO2 is reading above 93%, that patient may still need oxygen support [as this will depend if the patient is on supplemental oxygen or room air (which is an FiO2 of 21% oxygen)]! Using both your observations of the patient and the SPO2 percentage is necessary when choosing to provide oxygen therapy or not.
While arterial blood gas testing would provide the most accurate measurement of oxygenation in a patient, it may not be feasible to obtain. Testing does require a blood gas machine and strong technical skills; also, the restraint and arterial blood draw may cause discomfort in an awake or alert patient.
Once it has been verified the patient needs oxygen therapy, the method of delivery should be determined next.
METHODS OF DELIVERY
Flow-by oxygen
Flow-by oxygen is a non-invasive technique of providing oxygen supplementation. It can be provided during initial triaging and work-up, and can provide up to 25-40% FiO2 when held approximately 2 cm from the patient’s nose. Most clinics are able to provide this type of supplementation as the only equipment needed is an oxygen tank and oxygen tubing. While flow-by is a great choice to start the patient’s treatments, it is not good for long-term supplementation as it is considered wasteful (as oxygen escapes into the surrounding room), and the patient is most likely not going to stay in one spot for the duration of their treatment.

Photo courtesy of Melanie Newton, CVT VTS (ECC)
Face mask
Similar to using flow-by oxygen, using an anesthesia face mask is a non-invasive way to provide short-term oxygen supplementation. If using a tight fitting mask, an FiO2 of about 50-60% can be achieved when using a flow rate of 200 mL/kg/min or higher. However, most awake and alert patients find a tight-fitting mask to be distressing. Using a loose fitting mask may be less stressful, but may not achieve as high of an FiO2. Face mask supplementation cannot be realistically used long-term, as the patient is most likely going to move their face out of the mask if left alone. This is another method that can be used during triage, work-up situations, or patients under sedation, but isn’t the best long-term solution for oxygen therapy.
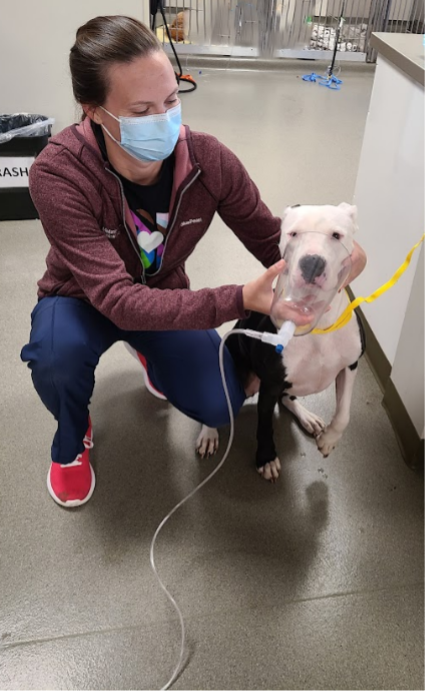
Photo courtesy of Melanie Newton, CVT VTS (ECC)
Oxygen cage
Oxygen cages are commercially available and typically found in larger emergency and specialty hospitals. They can also be made by using human pediatric incubators or a regular metal kennel with a Plexiglas door. An FiO2 of 50-60% (or higher) can be maintained in an oxygen cage. These cages allow the patient to move freely while still receiving oxygen supplementation. While larger oxygen cages can typically control temperature and humidity (such as Snyder cages), smaller cages or incubators may lead to hyperthermia due to low ventilation. Ice packs can be placed inside the cage in an effort to maintain a comfortable environment for the patient. An oxygen cage is non-invasive, and may be the most comfortable option for the patient. It should be noted, however, that accessing the patient for assessments and treatments will require opening the door to the cage, which will drop the FiO2 back to room air quickly.
Oxygen hood
In locations that do not have oxygen cages available but need a longer term, non-invasive solution for oxygen administration, an oxygen hood may be a good option. These can be built using cling wrap and an Elizabethan collar. Cling wrap is placed over most of the front of the cone, leaving a small area at the top or bottom for ventilation purposes (e.g., to blow out carbon dioxide). Oxygen tubing is then fed into the collar at the neck and secured to the side to prevent dislodging. This technique can reach an FiO2 of 30-40% if oxygen is delivered at a flow rate of 1-2 L/min. While this is a great option and does not require expensive equipment, the patient can still be at risk for hyperthermia as the temperature inside the hood cannot be controlled. Rarely, the patient can be at risk for suffocation if the plastic wrap occludes their nares or mouth.
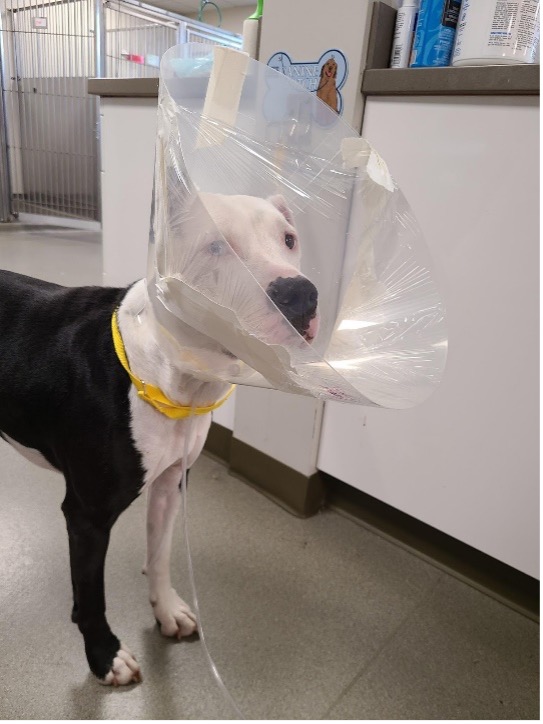
Photo courtesy of Melanie Newton, CVT VTS (ECC)
Nasal oxygen
In patients that require oxygen supplementation long term, nasal oxygen supplementation can be considered. An FiO2 of up to 70% can be achieved via nasal oxygen; however, this varies greatly based on flow rate and the patient’s respiratory rate. Nasal oxygen supplementation is given by placing red-rubber catheters or nasal cannulas into the nasal cavity. The catheter is measured to the medial canthus and then sutured to the patient’s nose to remain in place. Multiple side ports (or holes) can be used to allow oxygen to exit from more than the distal tip. Oxygen can be given through the catheter at flow rates of up to 100mL/kg/min. While not considered invasive, this technique can be uncomfortable for some patients. Topical 2% lidocaine or proparacaine should be provided into the nasal cavity prior to placement to ease distress. A humidification source has to be used when administering oxygen through this technique in order to avoid mucosal damage within the nasal cavity. A study performed in 2002 (Dunphy et. al 2002) showed that a unilateral nasal catheter can achieve proper oxygen supplementation; however, bilateral nasal catheters may increase patient comfort by allowing the flow rate to be lower while still achieving the desired FiO2. Nasal catheters allow for continual oxygen supplementation, even during patient assessments and treatments. Therefore, this method may be beneficial for the patient who cannot tolerate drops in FiO2 (such as an oxygen cage door being opened).
High flow nasal oxygen
High flow nasal oxygen is relatively new to veterinary medicine, but has been used with success. Oxygen is delivered via specialized nasal prongs at a rate of 0.4-2 L/kg/min, with the ability to provide up to 100% FiO2. A large benefit to this method is continuous positive airway pressure (often called CPAP), which has been shown to reduce the patient’s work of breathing and oxygenation (Jagodich et. al 2019). This technique does require specialized equipment, therefore may only be available in certain hospitals. High flow nasal oxygen can be used if the patient fails to improve with traditional oxygen therapy techniques, but does not need mechanical ventilation yet. A small disadvantage to high flow nasal oxygen is that some patients do find the high flow rate uncomfortable and may require sedation to prevent distress. The benefits of high flow nasal oxygen and the risks of sedation should be weighed on a case by case basis.
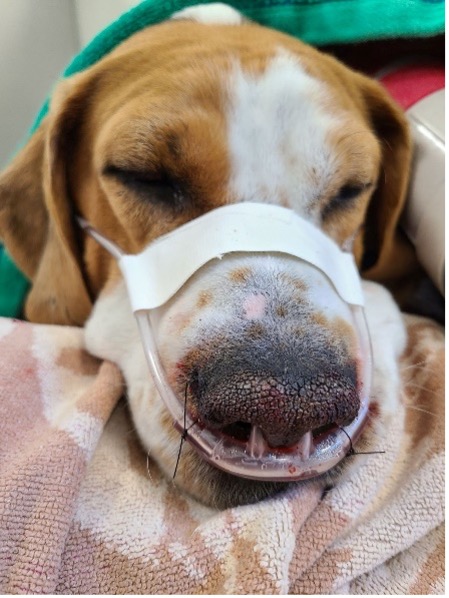
Photo courtesy of Melanie Newton, CVT, VTS (ECC)
Intubation and positive pressure ventilation (PPV)
Failure to improve with traditional oxygen supplementation techniques may result in a patient needing intubation, with or without positive pressure ventilation. By intubating the patient, oxygen can be delivered at 100% FiO2. Breaths can be provided to the patient, as well as positive end expiratory pressure (PEEP) if needed. Intubation supplies are relatively low-cost and positive pressure ventilation can be achieved using an ambu-bag, anesthesia machine, or mechanical ventilator, making this technique feasible in most clinics or hospitals. However, as this is the most invasive technique for delivering oxygen, sedation or anesthesia is required to maintain the delivery of oxygen via the endotracheal tube.
Oxygen supplementation is commonly used in veterinary practices for a wide variety of reasons. Using a hands-off approach, observation, and a pulse oximeter may be beneficial in determining the patient’s need for oxygen. The oxygen therapy technique will be determined by equipment available and the patient’s tolerance level. Overall, oxygen therapy should be used to improve the patient’s outcome, without causing distress.
References/Resources:
1. Dunphy ED, Mann FA, Dodham JR, et al. Comparison of unilateral versus bilateral nasal catheters for oxygen administration in dogs. J Vet Emerg Crit Care 2002;12:245-251.
2. Drobatz, Kenneth J., et al. “Oxygen Therapy.” Textbook of Small Animal Emergency Medicine, vol. 2, Wiley, Hoboken, NJ, 2019, pp. 1177–1182.
3. Engelhardt M, Crowe D. Comparison of six non-invasive supplemental oxygen techniques in dogs and cats. J Vet Emerg Crit Care 2004;14: S1-S17. https://doi.org/10.1111/j.1476-4431.2004.t01-25-04035.x
4. Jagodich TA, Bersenas AM, Bateman SW, Kerr CL. Comparison of high flow nasal cannula oxygen administration to traditional nasal cannula oxygen therapy in healthy dogs. J Vet Emerg Crit Care 2019; 29: 246–255. https://doi.org/10.1111/vec.12817
5. Jagodich, TA, Bersenas, AM, Bateman, SW, Kerr, CL. High-flow nasal cannula oxygen therapy in acute hypoxemic respiratory failure in 22 dogs requiring oxygen support escalation. J Vet Emerg Crit Care 2020;30:364–375. https://doi.org/10.1111/vec.12970
6. Silverstein, Deborah C., et al. “Oxygen Therapy.” Small Animal Critical Care Medicine, 3rd ed., Elsevier, St. Louis, MI, 2023, pp. 85–88.
Only VETgirl members can leave comments. Sign In or Join VETgirl now!
this was very informational. in my clinic setting we see alot of patients that’s need oxygen and I have never heard/ seen the use of the E-collar before.
Very informative. I didn’t realize there were so many different options.